Biotensegrity and the mechanics of fascia
Stephen M Levin, Graham Scarr
In: Schleip R, Stecco C, Driscoll M, Huijing P, (Eds.), 2022 Fascia: the tensional network of the human body., 2nd ed., Elsevier; pp. 232-238.
Summary
Biotensegrity recognizes that complex living structures result from interactions between some basic principles of self-organization and that these apply to everything from the smallest of molecules to the complete organism. Each one is a physical representation of the invisible forces that led to its formation and continue to flow through it. It thus appreciates the fascia as a continuous, heterarchical tension system containing multi-level sub-systems (compartments) under compression, with the mechanics described through a complex system of closed-chain kinematics operating within a global tensegrity configuration at every level. An appreciation of biotensegrity increases our understanding of what is taking place within the body and thus has the potential to improve therapeutic protocols and movement training.
Key Point 1: Biological structures emerge from interactions between some basic principles of self-organization.
Key Point 2: Fascia is a tensioned network encapsulating a complex heterarchical system of tissue compartments under compression.
Key Point 3: The mechanics of fascia are based on a body-wide system of closed-chain kinematics operating within a tensegrity configuration at every level.
Chapter contents: Introduction; Biotensegrity; The tensegrity model; The mechanics of fascia.
Introduction
Fascia is the fabric of the body; not the vestments covering the corpus but the material that gives it form: the warp and the weft. Muscle and bone, liver and lung, gut and urinary, brain and endocrine are all embroidered into the fascial web. It is a continuum, a tissue that emerges from the simple-celled embryo and evolves into a complex structural heterarchy that pervades the entire organism (Simon, 1962); from the interstitium that surrounds virtually every cell in the body to the extracellular matrix and fascial network with all their specialized variations (see chapter by Guimberteau). In fact, all the ‘connective tissues’ developed out of the embryonic mesenchyme, which means that the structures we label as muscles, bones, tendons, ligaments and fascia etc. are all derived from what is essentially the same tissue. As a result, it is likely that they share some similarities in their behavior with biotensegrity currently representing the most comprehensive approach to explaining this: the mechanics of fascia.
Biotensegrity
The biotensegrity concept recognizes that all living structures result from interactions between some basic principles of self-organization, or rules of physics, and that that these apply to everything from the smallest of molecules to the complete organism (Key Point 1) (Levin, 2015; Martin, 2016; Scarr, 2018). At the most basic level, atoms interact with each other in different ways that can be categorized as those that attract and those that repel (van der Waal’s forces, covalency, hydrogen-bonding and steric-repulsion etc.), and they spontaneously organize themselves into the most stable and energy-efficient configurations to form rigid crystals and flexible molecules. Each one develops as a distinct structural entity and effectively becomes a physical representation of the invisible forces that hold it together.
These structures form as they do because they are constrained by some basic principles of self-organization – geodesic geometry, close-packing and minimal-energy – and as attraction and repulsion always operate in conjunction with each other, they enable dynamic systems to automatically move towards a lower and more stable state of energy whenever possible. The same principles also apply at higher size-scales where we now refer to these invisible forces as tension and compression and are able to physically model their interactions (Connelly and Back, 1998).
The tensegrity model
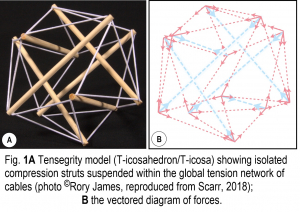 Fig. 1A shows a tensegrity structure consisting of six compression struts suspended within a tensioned network of cables, where the reciprocal action of these forces balances and maintains its particular shape. Each structural element is either under tension or compression, which means that tensegrity models are also displays of the invisible forces that hold them together: a three-dimensional ‘diagram of forces’ in mechanical terms (Fuller, 1975; Heartney, 2009) (Fig. 1B).
Fig. 1A shows a tensegrity structure consisting of six compression struts suspended within a tensioned network of cables, where the reciprocal action of these forces balances and maintains its particular shape. Each structural element is either under tension or compression, which means that tensegrity models are also displays of the invisible forces that hold them together: a three-dimensional ‘diagram of forces’ in mechanical terms (Fuller, 1975; Heartney, 2009) (Fig. 1B).
They are geodesics because their structural elements transfer these forces over the shortest distance; they are close-packed because their component parts automatically get as close as they can to each other; and they naturally balance in the simplest state of minimal energy. Such models are strong, light in weight, flexible and resilient. They can change shape with the minimum of effort and always return to the same position of stable equilibrium (dynamic stability), and their physical properties are very similar to living tissues. Each part is mechanically integrated with all the others so that when one part moves, everything is involved thus enabling forces, power and information to be transferred from one part of the system to another in a controlled way.
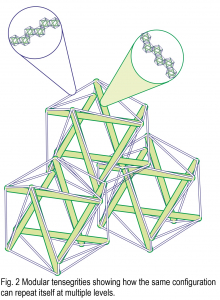 To add to this, each cable and strut could itself be constructed from a modular chain of smaller, similar tensegrities with each individual part maintaining its own structural role within the global heterarchy (Fig. 2). Such close-packed configurations are intrinsically stable, they optimize the load bearing ability and provide an intrinsic mechanism for distributing potentially damaging stresses throughout the structure (Connelly & Back, 1998). They are also ubiquitous in biology, where each ‘part’ is made from smaller parts, smaller parts, smaller parts… with each one nested and integrated within all those surrounding it to form a complete structural and functional unit (Reilly and Ingber, 2018). So, although living organisms appear to be highly complicated, both the simple tensegrity model and complex anatomy are formed through the combined actions of these physical forces and the same principles of self-organization, as outlined by Thompson (1917); but there is a problem.
To add to this, each cable and strut could itself be constructed from a modular chain of smaller, similar tensegrities with each individual part maintaining its own structural role within the global heterarchy (Fig. 2). Such close-packed configurations are intrinsically stable, they optimize the load bearing ability and provide an intrinsic mechanism for distributing potentially damaging stresses throughout the structure (Connelly & Back, 1998). They are also ubiquitous in biology, where each ‘part’ is made from smaller parts, smaller parts, smaller parts… with each one nested and integrated within all those surrounding it to form a complete structural and functional unit (Reilly and Ingber, 2018). So, although living organisms appear to be highly complicated, both the simple tensegrity model and complex anatomy are formed through the combined actions of these physical forces and the same principles of self-organization, as outlined by Thompson (1917); but there is a problem.
Orthodox biomechanics
Current biomechanical theory is based on the laws of classical mechanics as formulated by Galilei, Newton and Hooke etc., and applied to the behaviour of man-made machines from the Seventeenth Century (Borelli, 1680), and it has remained largely unchanged ever since. The problem is that these ‘rules’ were discovered on inanimate objects and they don’t necessarily apply to the non-linear dynamics of living tissues (Figure 3).
For example, biomechanics has taken the more ‘vertical’ part of the stress/strain curve to be a straight-line purely because this simplifies the analysis, but it is a fudge!
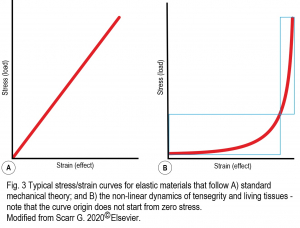 “True linearity does not exist in biology – not even as an approximation. Such a living system that is so profoundly complex at all its different levels and size-scales (10-9 – 101 m) cannot behave in a linear way – it is fundamentally impossible” (Blyum, 2020). In addition, the non-linear curve never reaches zero stress because of the intrinsic tension contained within the system (Schleip & Klingler, 2019). From a biotensegrity perspective, it is much more likely that biological structures operate within the more ‘horizontal’ low-stress/strain region where they will be more responsive, consume less energy and operate more efficiently (Levin, 2015).
“True linearity does not exist in biology – not even as an approximation. Such a living system that is so profoundly complex at all its different levels and size-scales (10-9 – 101 m) cannot behave in a linear way – it is fundamentally impossible” (Blyum, 2020). In addition, the non-linear curve never reaches zero stress because of the intrinsic tension contained within the system (Schleip & Klingler, 2019). From a biotensegrity perspective, it is much more likely that biological structures operate within the more ‘horizontal’ low-stress/strain region where they will be more responsive, consume less energy and operate more efficiently (Levin, 2015).
The lever-theory of joint motion (Borelli, 1680) also presents a problem because such mechanisms inherently generate bending moments, shear-stresses and potentially damaging stress-concentrations (this is not the case in tensegrity configurations), and it is most unlikely that developing tissues would be able to withstand the disastrous consequences of these (Levin, 2015). In addition, bones do not revolve around the fixed fulcrum of a lever, the mechanical behavior of cadavers is not the same as in living tissues, and the role of connective tissues is frequently ignored in the analysis. The nervous system is also incapable of controlling the huge complexity of joint movements on its own, all of which suggests that something else must be going on (Cabe, 2019).
These problems have persisted simply because there has been nothing else to explain them, but they have led biomechanics down a blind alley from which it is only just starting to recover. There is nothing wrong with classical mechanics, but it should be recognized that its value in a biological context is questionable because of the kinematic indeterminacy inherent within the latter; Biotensegrity now offers a more comprehensive perspective that resolves all these issues (Table 1).
Table 1
Comparison of properties of biologic systems, lever mechanics and tensegrity icosahedron
Biologic systems Lever systems Tensegrity icosahedron
Non-linear Linear Non-linear
Global Local Global
Structurally continuous Discontinuous Structurally continuous
Gravity independent Gravity dependent Gravity independent
Omnidirectional Unidirectional Omnidirectional
Low energy High energy Low energy
Flexible joints Rigid joints Flexible joints
The mechanics of fascia
A popular misconception about the structure of the human body, and biotensegrity, is that it is simply a system of compressed bones suspended within a tensioned network of muscles and fascia, but this view is far too simplistic. Anatomy is NOT a collection of isolated bits that operate in a local piecemeal-like way but a complex, fully integrated, nested modular arrangement where the function of each part is dependent on all those surrounding it (Fig. 2).
Fascia is a continuous body-wide architectural system containing multi-level sub-systems, or compartments, that ultimately surround individual cells (Key Point 2). Within the higher-level compartments are bones, muscles, lungs, heart, liver and bladder etc. Smaller fascial compartments enclose glands, blood vessels, nerves and other tissues, while individual muscle cells are encapsulated within tubular endomysial, perimysial and epimysial compartments.
All these different ‘compartments’ are regional specializations of what is essentially the same tissue with each one playing a subtly different role. The parenchymal cells within them provide the ‘power’ (myofiber, cardiac, hepatic, nerve and adipose etc.) while the fascia contains the cells and supports their functions. From an embryological, biotensegrity and logical perspective, bones must then also be considered as compartmental specializations of fascia (mineralized but still flexible) with their own parenchymal cells (osteocytes, osteoblasts and osteoclasts) (Bordoni and Lagana, 2019; Levin, 2018).
The fascia is also continuous with tendons, ligaments, aponeuroses and membranes etc., as it is within every bone and organ in the body, so that at the microscopic level there is no clear distinction between where one ends and the other begins (see chapter by Guimberteau). It is intrinsically tensioned (Schleip & Klingler, 2019) but cannot operate without a compressional component, which is neatly provided by the cells, fluid-bound proteoglycans and other structures within each ‘compartment’. Muscles are then not so much ‘organs of movement’ but rather dynamic tensioners of the system. The ‘mechanics of fascia’ is thus something of a misnomer because the same principles that led to its formation also express themselves through a unified mechanical system that includes every part of the body.
Closed-chain kinematics
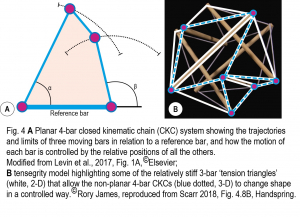 Closed kinematic chains (CKCs) have been used in mechanical engineering for hundreds of years because they provide a simple and efficient mechanism with multiple applications; and are also well-described in biology (Levin et al., 2017). Closed-chain kinematics couple multiple parts into continuous mechanical loops with each one influencing the position, motion and stability of all the others in the system and enabling the controlled transfer and amplification (or attenuation) of force, speed and kinetic energy. They also describe the basic mechanics of tensegrity (Key Point 3) (Fig. 4).
Closed kinematic chains (CKCs) have been used in mechanical engineering for hundreds of years because they provide a simple and efficient mechanism with multiple applications; and are also well-described in biology (Levin et al., 2017). Closed-chain kinematics couple multiple parts into continuous mechanical loops with each one influencing the position, motion and stability of all the others in the system and enabling the controlled transfer and amplification (or attenuation) of force, speed and kinetic energy. They also describe the basic mechanics of tensegrity (Key Point 3) (Fig. 4).
Kinematics is all about the ‘geometry of motion’, and the simplest arrangement that enables the structure itself to control this is the planar 4-bar, where the length and position of each linkage bar determines the mechanical behaviour of all the others in the system, and the changing angular relationships between them (e.g. α and β) define its characteristic non-linear properties. In comparison, 3-bar triangular shapes can be relatively rigid and are important because of their stability, while those with five or more bars can be uncontrollable on their own but are crucial to more complicated systems. Note that this mechanical system is not synonymous with exercise regimes that take a similar name.
At first sight, these closed-chain linkages might appear to be just complex lever systems, but this is not the case in a biological context where every bar and connection between them (‘pin-joint’) consists of multiple structures acting within their own CKC sub-systems (Fig. 2). Each one will be active in either tension or compression (at that particular size-scale) and evolutionarily developed in response to the forces flowing through it. There is thus no true fulcrum or bending-moment around which a lever can act, and ‘automatic shifting-suspension mechanism’ might be a better description.
The mechanics of CKCs in a biotensegrity context apply equally well to the omnidirectional interactions within and between different molecules, cells, tissues (e.g., fascia), joints and the whole body, with their non-linear responses intrinsically related to the heterarchical geometry (Fig. 3B). Like a pantograph, they enable such systems to amplify (or attenuate) force, speed and kinetic energy, and thus operate much faster than muscles can contract with the structure itself guiding motion. As the positional status of each ‘joint’ is dependent on the interactions between a vast number of different fascia-linked tissues, the effect of every muscle contraction can then become distributed over a wide anatomical field. Conversely, multiple muscles can transfer and concentrate their power through the CKC system and cause movement patterns that might otherwise seem impossible.
Such configurations allow the body to respond instantly to rapidly changing conditions and regulate complex movements in ways that are beyond the sole capability of the nervous system (Cabe, 2019). Where control and the processing of information (force and direction) are embedded within the structure itself and allow the system to perform its own logic computations in synergy with the neurology. CKC arrangements also have distinct evolutionary advantages as they enable multiple mechanical functions to become optimized throughout the entire system during embryological development and provide a mechanism for increasing diversity (Levin et al., 2017; Scarr, 2018).
Biotensegrity recognizes that living organisms are physical representations of a complex balance of invisible force vectors, and that it is the efficient separation of tension and compression into different structural modules (bars) that enables them to change shape with the minimum of effort and remain completely stable throughout (dynamic stability). CKC mechanics enable the structural system to efficiently distribute forces over a wide variety of different pathways and compensate for any deficits that arise within the system. As a degenerate system, it would also enable the co-operation of fundamentally different components to produce consistently reliable outputs under diversely fluctuating conditions (Wilson and Kiely, 2016).
Whenever nature uses the same strategy in a variety of different situations there is probably an underlying energetic advantage to its appearance, and embryological development and evolution consistently favor those patterns and shapes that are the most efficient in terms of stability, materials and mass. Biology is not dependent on those contrived laws that govern the behaviour of man-made machines (e.g. lever theory) but conforms more to the newly-emerging physics of soft-matter – the characteristics and behavior of bubbles, foams, colloids, polymers and liquid-crystals etc. (Hirst,2013). Biotensegrity now reverses the centuries-old concept of the skeleton as the ‘frame upon which the soft tissue is draped’ and replaces it with an integrated, dynamic and multi-level fascial heterarchy fit for 21st Century biomechanics.
References
Blyum L. 2020. The unreasonable effectiveness of light touch. In: Trewartha JE, Wheeler SL, editors. Scars, adhesions and the biotensegral body: science, assessment and treatment Edinburgh: Handspring; 47-63.
Bordoni B, Lagana MM. 2019. Bone tissue is an integral part of the fascial system. Cureus 11: e3824
Borelli GA. 1680. De Motu Animalium. Lugduni batavorum, Petrum Vander As.
Cabe PA. 2019. All perception engages the tensegrity-based haptic medium. Ecological Psychology 31(1): 1-13.
Connelly R, Back A. 1998. Mathematics and tensegrity. American Scientist, 86: 142-151.
Fuller RB. 1975. Synergetics: explorations in the geometry of thinking. London: Collier Macmillan.
Heartney E. 2009. Kenneth Snelson: forces made visible. Massachusetts: Hard Press Editions.
Hirst LS. 2013. Fundamentals of soft matter science. Boca Raton, Florida: CRC Press, Taylor & Francis Group.
Levin SM. 2015. Tensegrity: the new biomechanics. In: Hutson M, Ward A., editors. Textbook of musculoskeletal medicine., 2nd edition. Oxford: Oxford University Press.
Levin SM. 2018. Bone is fascia. Available Online at: https://www.researchgate.net/publication/327142198_Bone_is_fascia
Levin SM, Lowell de Solórzano S, Scarr G. 2017. The significance of closed kinematic chains to biological movement and dynamic stability. Journal of Bodywork and Movement Therapies 21: 664-72.
Martin DC. 2016. Living biotensegrity: interplay of tension and compression in the body. Munich: Keiner.
Reilly CB, Ingber DE. 2018. Multi-scale modeling reveals use of hierarchical tensegrity principles at the molecular, multi-molecular, and cellular levels. Extreme Mechanics Letters 20: 21-8.
Scarr G. 2018. Biotensegrity: the structural basis of life. 2nd edition Edinburgh: Handspring.
Scarr G. 2020. Biotensegrity: what is the big deal? Journal of Bodywork and Movement Therapies 24:134-137.
Schleip R, Klingler W. 2019. Active contractile properties of fascia. Clinical Anatomy 32: 891-895.
Simon HA. 1962. The architecture of complexity. Proceedings of the American Philosophical Society 106: 467-82.
Thompson D. 1917. On growth and form. Cambridge: Cambridge University Press.
Wilson J, Kiely J. 2016. The multi-functional foot in athletic movement: extraordinary feats by our extraordinary feet. Human Movement 17: 15-20.
e to add your own text
