Cranial Vault
A MODEL OF THE CRANIAL VAULT AS A TENSEGRITY STRUCTURE AND ITS SIGNIFICANCE TO NORMAL AND ABNORMAL DEVELOPMENT
International Journal of Osteopathic Medicine
2008;11:80-89 link
Graham Scarr request copy
Abstract
Traditional views of the human cranial vault are facing challenges as researchers find that the complex details of its development do not always match previous opinions that it is a relatively passive structure. In particular, that stability of the vault is dependent on an underlying brain; and sutural patency merely facilitates cranial expansion. The influence of mechanical forces on the development and maintenance of cranial sutures is well-established, but the details of how they regulate the balance between sutural patency and fusion remain unclear. Previous research shows that mechanical tensional forces can influence intra-cellular chemical signalling cascades and switch cell function; and that tensional forces within the dura mater affect cell populations within the suture and cause fusion.
Understanding the developmental mechanisms is considered important to the prevention and treatment of premature sutural fusion – synostosis – which causes skull deformity in approximately 0.05% of live births. In addition, the physiological processes underlying deformational plagiocephaly and the maintenance of sutural patency beyond early childhood require further elucidation.
Using a disarticulated plastic replica of an adult human skull, a model of the cranial vault as a tensegrity structure which could address some of these issues is presented.
The tensegrity model is a novel approach for understanding how the cranial vault could retain its stability without relying on an expansive force from an underlying brain, a position currently unresolved. Tensional forces in the dura mater have the effect of pushing the bones apart, whilst at the same time integrating them into a single functional unit. Sutural patency depends on the separation of cranial bones throughout normal development, and the model describes how tension in the dura mater achieves this, and influences sutural phenotype. Cells of the dura mater respond to brain expansion and influence bone growth, allowing the cranium to match the spatial requirements of the developing brain, whilst remaining one step ahead and retaining a certain amount of autonomy. The model is compatible with current understandings of normal and abnormal cranial physiology, and has a contribution to make to a hierarchical systems approach to whole body biomechanics.
A follow-up project with three co-authors to produce an animated simulation of the skull as a tensegrity structure was presented at the International Fascia Research Congress in Montreal in 2022, and can be viewed here.
Introduction
 For many years it has been widely accepted that the cranial vault expands through an outward pushing pressure from the growing brain, with the sutures merely accommodating its growth and fusing in the third decade of life.1,2 However, recent data suggests that daily brain growth is too small to induce sutural osteogenesis, and that in any case, substantial growth is over before the completion of sutural growth.3,4,5,6 Human facial sutures normally remain patent until at least the seventh or eighth decade, whereas the timing of sutural fusion in the cranial vault is extremely variable and unreliable forensically.7,8 Many factors affect cranial enlargement – some are genetic while others are epigenetic.
For many years it has been widely accepted that the cranial vault expands through an outward pushing pressure from the growing brain, with the sutures merely accommodating its growth and fusing in the third decade of life.1,2 However, recent data suggests that daily brain growth is too small to induce sutural osteogenesis, and that in any case, substantial growth is over before the completion of sutural growth.3,4,5,6 Human facial sutures normally remain patent until at least the seventh or eighth decade, whereas the timing of sutural fusion in the cranial vault is extremely variable and unreliable forensically.7,8 Many factors affect cranial enlargement – some are genetic while others are epigenetic.
Understanding the developmental mechanisms of the cranium is considered important to the prevention and treatment of the pathologies affecting the neonatal cranium. Craniosynostosis is the premature fusion of one or more of the cranial sutures resulting in skull deformity, and occurs in roughly 1 in 2000 live births.4 It may be associated with specific genetic syndromes or occur sporadically, and any cranial suture may be involved, although with differing frequencies.2,9,10 Premature fusion results in arrested bone growth perpendicular to the synostosed suture, with subsequent abnormal compensatory growth in the patent sutures.1,2,9,11 Another skull deformity, not due to synostosis, is positional moulding or deformational plagiocephaly. When present at birth it is the result of in-utero or intrapartum molding, often associated with multiple births, forceps or vacuum-assisted delivery; or post-natally resulting from a static supine positioning.12 One of the difficulties during this period is differentiating premature fusion from abnormal moulding. By the time children are diagnosed with craniosynostosis, the suture has already fused and the associated dysmorphology well established. Surgical intervention may then be necessary for neurological or cosmetic reasons.
 The adult skeleton is mostly capable of healing defects and deficiencies via the formation of new bone. However, while children under the age of 2 years maintain the capacity to heal large calvarial defects, adults are incapable of healing the smallest of injuries. The coordinating mechanisms behind normal and abnormal development are currently incomplete,10,13 and the model to follow presents a novel approach to furthering our understanding of the processes involved. Although many readers will have an extensive knowledge of the cranium, others may be unfamiliar with the details which underlie the significance of this model, and a brief overview follows.
The adult skeleton is mostly capable of healing defects and deficiencies via the formation of new bone. However, while children under the age of 2 years maintain the capacity to heal large calvarial defects, adults are incapable of healing the smallest of injuries. The coordinating mechanisms behind normal and abnormal development are currently incomplete,10,13 and the model to follow presents a novel approach to furthering our understanding of the processes involved. Although many readers will have an extensive knowledge of the cranium, others may be unfamiliar with the details which underlie the significance of this model, and a brief overview follows.
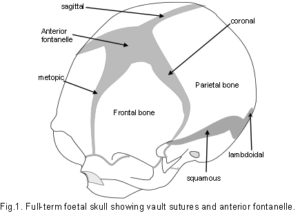
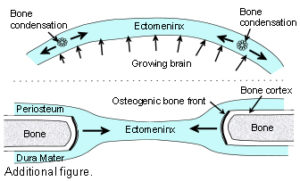
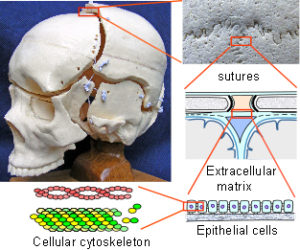
The Cranial Vault or calvarium: The cranial vault, or calvarium, surrounds and encloses the brain, and is formed from several plates of bone which meet at sutural joints, unique to the skull, and which display a variety of morphologies specific to each suture.2,7,11,14,15 The high compressive and tensile strength of bone provides mechanical protection for the underlying brain, while the sutural joints provide a soft interface and accommodate brain growth.10 The vault bones are the frontal, parietals and upper parts of the occiput, temporals and sphenoid. Inferior to the vault is the cranial base, or chondrocranium, which is made up of the lower parts of the occiput and temporals, the ethmoid and the majority of the sphenoid. In the embryo, the vault bones develop through ossification of the ectomeninx – the outer membranous layer surrounding the brain; while the cranial base develops through an additional cartilaginous stage,2, 16 the significance of which will be discussed later (Individual bones spanning both regions fuse at a later stage). Enlargement of the neurocranium occurs through ossification of sutural mesenchyme at the bone edges, and an increase in bone growth around their perimeters.1,15 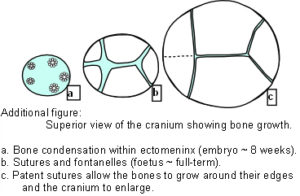 During this process, the ectomeninx becomes separated by the intervening bones into an outer periosteum and internal dura mater. By the time of full term birth, the growth of the different bones has progressed sufficiently so that they are in close apposition, only separated by the sutures which intersect at the fontanelles (Figure 1). At full-term birth, sutural bone growth is progressing at about 100 microns/day, but this rate rapidly decreases after this. Maintenance of sutural patency is essential throughout for normal development of the brain and craniofacial features.2,4,10 The brain has usually reached adult size by the age of 7 years but the sutures normally persist long after this – until at least 20 years of age. Even after this, there is considerable variation in the pattern and timing of sutural fusion in the human adult throughout life.2,7,8,16 Animal studies of the cranial vault clearly demonstrate sutural patency throughout.2,16
During this process, the ectomeninx becomes separated by the intervening bones into an outer periosteum and internal dura mater. By the time of full term birth, the growth of the different bones has progressed sufficiently so that they are in close apposition, only separated by the sutures which intersect at the fontanelles (Figure 1). At full-term birth, sutural bone growth is progressing at about 100 microns/day, but this rate rapidly decreases after this. Maintenance of sutural patency is essential throughout for normal development of the brain and craniofacial features.2,4,10 The brain has usually reached adult size by the age of 7 years but the sutures normally persist long after this – until at least 20 years of age. Even after this, there is considerable variation in the pattern and timing of sutural fusion in the human adult throughout life.2,7,8,16 Animal studies of the cranial vault clearly demonstrate sutural patency throughout.2,16
The Dura Mater: The dura mater is the outer one of three membranes surrounding the brain (fig. 2). Its outer surface – the endosteal layer, is loosely attached to most of the inner bone surface, particularly in children, but more firmly attached around the bone margins, the base of the skull and foramen magnum. The inner meningeal layer of the dura mater continues down through the foramen magnum and surrounds the spinal cord as far as the sacrum. This layer also reduplicates inwards as four sheets which partially divide the cranial cavity and unite along the straight sinus – the falx cerebri, falx cerebellum and bilateral tentorium cerebelli.
The internal structure of the dura mater consists of inner and outer elastic networks and integumentary layers, and a collagen layer; although abrupt boundaries between these ‘layers’ cannot be distinguished histologically.17 The collagen layer occupies over 90% of its thickness, with collagen fibres arranged in parallel bundles and differing orientations – varying from highly aligned to apparently random, and arranged in lamellae.18 Typically, with age, the dura mater thickness changes from 0.3 to 0.8 mm.17,18 Collagen has the strongest mechanical properties of the different structural proteins, and fibre orientation has been observed to coincide with the direction of tensile stress.9,18,19,20
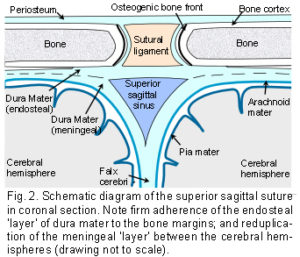 The Sutures: Adjacent cranial vault bones are linked through fibrous mesenchymal tissue, referred to as the sutural ligament (fig. 2).15 The two layers which derive from the embryonic ectomeninx – the periosteum and dura mater, continue across the suture, and also unite around the bone edges.15 In the cranial base, ossification occurs through cartilage precursors, some of which fuse together in the foetus or early childhood.
The Sutures: Adjacent cranial vault bones are linked through fibrous mesenchymal tissue, referred to as the sutural ligament (fig. 2).15 The two layers which derive from the embryonic ectomeninx – the periosteum and dura mater, continue across the suture, and also unite around the bone edges.15 In the cranial base, ossification occurs through cartilage precursors, some of which fuse together in the foetus or early childhood.
The synchondroses are the intervening cartilages between the bones of the cranial base. The spheno-basilar synchondrosis normally ossifies in the third decade, and the petro-occipital fissure (synchondrosis) in the seventh.21 The cranial base is relatively stable during development, with the greatest size changes taking place in the vault.
Morphogenesis and phenotypic maintenance of the sutures is a result of intrinsic differences within the dura mater.1,5,10,16,20,22 The significant factors in this are cellular differentiation, intercellular signals and mechanical signals.23
(1) Cells of the dura mater beneath the suture undergo epithelial-mesenchymal transitions – a mechanism for diversifying cells found in complex tissues, and migrate into the suture as distinct cell populations.23,24,25 Fibroblast-like cells in the centre produce collagen and maintain suture patency. Those with an osteoblast lineage also produce a collagen matrix, but lead onto bone formation at the suture margins, causing the cranial bones to expand around their perimeters.13 Osteoclast mediated bone resorption may be necessary for changes in the complex morphological characteristics at the sutures edges.26 A complex coupling between fibroblast, osteoblast and osteoclast populations determines the actual position and rate of sutural development.5,10,26,27 In addition, a critical mass of apoptotic cells within the suture is essential to maintaining the balance between sutural patency and new bone formation.10,14
(2) Intercellular signalling influences epithelial cell function through the production and interactions of soluble cytokines such as the ‘fibroblast growth factors’ and ‘transforming growth factors’.23,25 The cells at the approximating edges of the bones, either side of the suture (bone fronts), set up a gradient of growth factor signalling which regulates the sequential gene expression of other cells, and causes changes in the spatial and temporal development of different cell populations.10,13,22,28
(3) Mechanical signals.The morphology of the suture also reflects the intrinsic tensional forces in the dura mater, in the order of nano or pico Newtons.1,3,27,28 Regional differentials in this tension create mechanical stresses which interact and exert their effects on the cells, stimulating them to differentiate and produce different cell populations.4,20,23,27,28 The sensitivity of the cellular cytoskeleton to tensional forces, and the particular pattern of stress application, has been shown to be crucial in determining the cellular response through a process of mechanotransduction.2,28-34 Given that the cytoskeleton is attached to the surrounding extracellular matrix through mechano-receptors in the cell membrane, a mechanical force transfer between them can produce global changes within the cell by altering the cytoskeletal tension. Multiple chemical signalling pathways are activated within the cell as a result, and together with intercellular chemical signals, provides multiplexed switching between different functional states such as differentiation, proliferation and cell death.29,30,32
 It is actually not an essential requirement for a spherical tensional structure to be maintained through an expansive force (such as a growing brain) in order to remain stable.3,35 The proposal here is that the calvarium of the neonate could be such a structure which maintains its shape through other mechanisms, being influenced by the expanding brain as a secondary factor.
It is actually not an essential requirement for a spherical tensional structure to be maintained through an expansive force (such as a growing brain) in order to remain stable.3,35 The proposal here is that the calvarium of the neonate could be such a structure which maintains its shape through other mechanisms, being influenced by the expanding brain as a secondary factor.
THE TENSEGRITY MODEL
The concepts of tensegrity have become increasingly recognized over the last thirty years as a model for understanding some of the structural properties of living organisms.29,30,35-42 This appreciation follows from investigations in the 1940s by the sculptor Kenneth Snelson, and the architect Buckminster Fuller, into novel structures in free standing sculpture and building design.35,41 Although Snelson actually discovered the concept, and has used it to great effect in his sculptures, it was Fuller who defined the basic geodesic mathematics. The word ‘tensegrity’ is derived from the words ‘tension’ and ‘integrity’ and describes structures which are inherently stable as a result of their particular geometry.
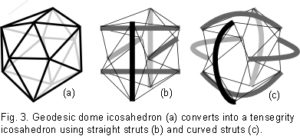
 Fuller found the icosahedron to be a useful model for describing certain aspects of geodesic geometry – the geodesic dome and tensegrity.35,36 The outstanding feature of geodesic domes is that they have a rigid external frame maintaining their shape, based on a repeating pattern of simple geometry (fig. 3a). In the human body, this type of structure is found in the cytoskeletal cortex of most cells;43 and in the erythrocyte, the geodesic structure is considered a primary contributor to the functionality of its peculiar shape.44 Tensegrity structures have been well described by Ingber in the inner cytoskeletons of cells;29,30 and Levin in the shoulder, pelvis and spine,36-40 suggesting their ubiquity throughout the organism.
Fuller found the icosahedron to be a useful model for describing certain aspects of geodesic geometry – the geodesic dome and tensegrity.35,36 The outstanding feature of geodesic domes is that they have a rigid external frame maintaining their shape, based on a repeating pattern of simple geometry (fig. 3a). In the human body, this type of structure is found in the cytoskeletal cortex of most cells;43 and in the erythrocyte, the geodesic structure is considered a primary contributor to the functionality of its peculiar shape.44 Tensegrity structures have been well described by Ingber in the inner cytoskeletons of cells;29,30 and Levin in the shoulder, pelvis and spine,36-40 suggesting their ubiquity throughout the organism.
In development of the model, the icosahedron is converted into a tensegrity structure by using six new compression members to traverse the inside, connecting opposite vertices and pushing them apart (fig. 3b). Replacing the edges with cables now results in the outside being entirely under isometric tension. 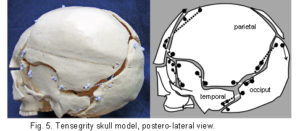 The inward pull of the cables is balanced by the outward push of the struts, providing structural integrity so that the compression elements appear to float within the tension network. A load applied to this structure causes a uniform change in tension around all the edges (cables), and distributes compression evenly to the six internal struts, which remain distinct from each other and do not touch.35 (Some of the edges of the geodesic dome (fig. 3a) have disappeared in the transition to tensegrity (fig. 3b) because they now serve no structural purpose and are redundant.) Replacing the straight struts (fig. 3b) with curved ones (fig. 3c) maintains the same stability, but they now surround a central space. In the same way, the curved struts can be replaced with curved plates (not shown) and the structure still retains its inherent stability.
The inward pull of the cables is balanced by the outward push of the struts, providing structural integrity so that the compression elements appear to float within the tension network. A load applied to this structure causes a uniform change in tension around all the edges (cables), and distributes compression evenly to the six internal struts, which remain distinct from each other and do not touch.35 (Some of the edges of the geodesic dome (fig. 3a) have disappeared in the transition to tensegrity (fig. 3b) because they now serve no structural purpose and are redundant.) Replacing the straight struts (fig. 3b) with curved ones (fig. 3c) maintains the same stability, but they now surround a central space. In the same way, the curved struts can be replaced with curved plates (not shown) and the structure still retains its inherent stability.
 The use of curved struts in tensegrity can be understood through structural hierarchies. In biology, it is common for component structures to be made up of smaller structures, which are themselves made up of still smaller substructures. Structural hierarchies provide a mechanism for efficient packing of components, dissipation of potentially damaging stresses and integration of all parts of the system. Thus, the appearance of curves at one scale are seen to result from interactions of components at a smaller scale, and the forces of tension (attraction) and compression (repulsion) always act in straight lines within them.
The use of curved struts in tensegrity can be understood through structural hierarchies. In biology, it is common for component structures to be made up of smaller structures, which are themselves made up of still smaller substructures. Structural hierarchies provide a mechanism for efficient packing of components, dissipation of potentially damaging stresses and integration of all parts of the system. Thus, the appearance of curves at one scale are seen to result from interactions of components at a smaller scale, and the forces of tension (attraction) and compression (repulsion) always act in straight lines within them.
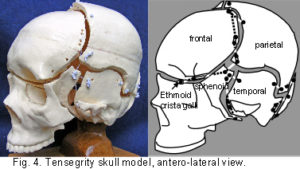
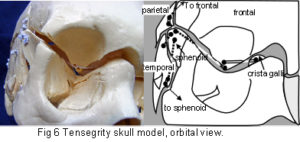
The plastic adult skull model illustrated in figures 4 – 7 shows curved plates of cranial bone – representing the compression struts, apparently ‘floating’ in the dura mater – shown here as elastic tension cords. The bones do not make actual contact with each other at any point. As this paper essentially concerns the cranial vault, the facial bones have not been separated. Bones of the cranial base are shown here as part of an overall tensioned structure, in spite of the synchondroses being under a certain amount of compression in vivo.
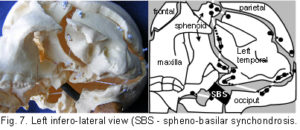 Their development in the early embryo could be part of a tensegrity structure, only changing to compression as the cartilage growth plates replace membrane between the bones. They are shown as they are in order to demonstrate the potential of the tensegrity principle through all stages of cranial development. Substituting the tension cords of these model sutures with a compression union would not alter that principle in the vault. The spheno-basilar synchondrosis (fig 7) has been distracted in order to display the isolation of each bone within the dura mater more clearly. (It also supports the unbalanced weight of the face; but see the additional wire model below.) Internal cranial structures have been omitted for the sake of clarity.
Their development in the early embryo could be part of a tensegrity structure, only changing to compression as the cartilage growth plates replace membrane between the bones. They are shown as they are in order to demonstrate the potential of the tensegrity principle through all stages of cranial development. Substituting the tension cords of these model sutures with a compression union would not alter that principle in the vault. The spheno-basilar synchondrosis (fig 7) has been distracted in order to display the isolation of each bone within the dura mater more clearly. (It also supports the unbalanced weight of the face; but see the additional wire model below.) Internal cranial structures have been omitted for the sake of clarity.
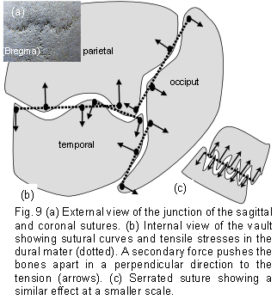
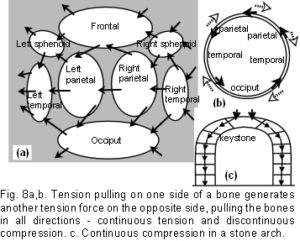
A fundamental characteristic of tensegrity structures is, as Fuller described it, “…continuous tension and discontinuous compression”.35 These concepts are illustrated in figure 8a which shows a schematic diagram of the bones spread out in two dimensions. The bones are the compression elements which are being pulled by dural tension (only a small number of tension forces pulling in one general direction are shown in this diagram). Here they remain distinct from each other and do not make contact with each other at any point – ‘discontinuous compression’. This contrasts with figure 8c, which shows the compressive load of a stone wall bearing down through the keystone and both sides of the arch – the compression force here is continuous.
 Returning to figure 8a, the tension cords are pulling in different directions, but a resultant tensional force develops (large arrows) which is dependent on the size and direction of the contributing tensions (the ‘parallelogram of forces’ in mechanics terminology). Starting with the left temporal: the tension pulls the left parietal (indirectly here) towards the left temporal in the direction of the resultant force. At the same time, the left parietal is pulling on the right parietal through the same mechanism, and this in turn is pulling on the right temporal. The consequence of all this is brought together in figure 8b, showing the same bones arranged in a circular anatomical sequence, the resultant tension pulling on each bone in turn, passing around the circle, and ultimately pulling on itself – ‘continuous tension’. Before running away with thoughts of perpetual motion, it must be pointed out that an equal and opposite tensional force will also be pulling in the opposite direction with the effect of – zero – nothing happens! This same isometric tension is acting across all the sutures in 3 dimensions, and because it is a tensegrity structure, the consequence is that all the tensional forces are balanced, the bones appear to float, and unless acted upon by another force, the structure will remain as it is. The precise placement and directions of the tensions is extremely important if the structure is to maintain itself as described, and is detailed later. While the simple 6-strut model is useful for demonstrating tensegrity, such structures can be made using any number of compression struts from two upwards, with the compression members remaining distinct from each other.45
Returning to figure 8a, the tension cords are pulling in different directions, but a resultant tensional force develops (large arrows) which is dependent on the size and direction of the contributing tensions (the ‘parallelogram of forces’ in mechanics terminology). Starting with the left temporal: the tension pulls the left parietal (indirectly here) towards the left temporal in the direction of the resultant force. At the same time, the left parietal is pulling on the right parietal through the same mechanism, and this in turn is pulling on the right temporal. The consequence of all this is brought together in figure 8b, showing the same bones arranged in a circular anatomical sequence, the resultant tension pulling on each bone in turn, passing around the circle, and ultimately pulling on itself – ‘continuous tension’. Before running away with thoughts of perpetual motion, it must be pointed out that an equal and opposite tensional force will also be pulling in the opposite direction with the effect of – zero – nothing happens! This same isometric tension is acting across all the sutures in 3 dimensions, and because it is a tensegrity structure, the consequence is that all the tensional forces are balanced, the bones appear to float, and unless acted upon by another force, the structure will remain as it is. The precise placement and directions of the tensions is extremely important if the structure is to maintain itself as described, and is detailed later. While the simple 6-strut model is useful for demonstrating tensegrity, such structures can be made using any number of compression struts from two upwards, with the compression members remaining distinct from each other.45
 The model was constructed from a full size plastic adult skull obtained from a medical suppliers and cut into the individual bones using a fine coping saw, with the exceptions of the facial bones which remain as a unit with the sphenoid. Although the intricacies of the serrate sutures could not be followed exactly, comparison with a real bone skull confirmed their essential similarities for the purpose described. Holes drilled at the bone perimeters were threaded with an elastic cord, as used in textile manufacture.The tension cords are positioned so that they illustrate the nature of the tensegrity structure and do not necessarily follow any particular anatomic structure. However, during positioning of the attachment holes, it became apparent that they should be as close to the edge as possible in order for the structure to work effectively. It was also evident that the various curves of the bone edges, in all three dimensions, facilitated a separation of the bones by making alternate attachments between the peaks of opposing bone edge convexities (fig 9a).
The model was constructed from a full size plastic adult skull obtained from a medical suppliers and cut into the individual bones using a fine coping saw, with the exceptions of the facial bones which remain as a unit with the sphenoid. Although the intricacies of the serrate sutures could not be followed exactly, comparison with a real bone skull confirmed their essential similarities for the purpose described. Holes drilled at the bone perimeters were threaded with an elastic cord, as used in textile manufacture.The tension cords are positioned so that they illustrate the nature of the tensegrity structure and do not necessarily follow any particular anatomic structure. However, during positioning of the attachment holes, it became apparent that they should be as close to the edge as possible in order for the structure to work effectively. It was also evident that the various curves of the bone edges, in all three dimensions, facilitated a separation of the bones by making alternate attachments between the peaks of opposing bone edge convexities (fig 9a).
DISCUSSION
One of the difficulties found in constructing this model was the unexpected vault shape changes caused by adjusting individual cord tensions. Tensegrity structures have visco-elastic properties similar to biological structures, and this can cause them to behave unpredictably because of a non-linear relationship between stress and strain.9,35,46 A summary of some of the significant mechanical aspects of tensegrity design and how they apply to the human skull follows:
3.1. Stability
Stability is achieved through the configuration of the whole network, and not because of the individual components. The model describes a mechanism whereby the calvarial shape could be maintained independently of any outward-pushing pressure from the brain within,1-6 a position currently unresolved. The sutures remain under tension (tension being necessary for regulating bone growth), while the bones remain mechanically distinct from the brain, being influenced through cells of the dura mater to expand. It is likely that the vault shape of the early foetus would be reliant on the expanding brain pushing outwards on the ectomeninx,2,4,10 but tensegrity could become a significant factor after 8 weeks, as ossification stiffens the membranous tissue and transfers tensional stresses across the developing bone (fig 8a).23 Chondrification would transform the base into a more ‘geodesic dome’ structure with greater stability (fig. 3a), and reorient certain vectors of growth influencing the greater expansion of the vault.1,2
During construction of the model, it became evident that it would only work effectively if the tension cords were attached near the edges of the bone. In children, the strongest attachments of the dura mater are also around the bone margins, suggesting that this may be significant and congruent with the mechanism being modelled.20 Continuity of dural tension is thus maintained beneath the bone and may affect intercellular signalling from one side to another. Firmer attachments of dura mater in the centre of adult bone would not affect the tensegrity principle, but implies a change in that signalling, and may influence the lack of bone healing capability in the skull after early childhood.
It must be emphasized that this model describes a structural mechanism which may be functioning in living tissue. It would not work in preserved skulls or cadavers where sutural and dural tissues have lost their elastic resiliency, and the structure becomes fixed under continuous compression (Figure 8c).
3.2. Balance
The tension and compression components are balanced mechanically throughout the entire structure, which will optimize automatically so as to remain inherently stable. The various curves of the bone edges, in all three dimensions, facilitate a separation of the bones through alternate tension attachments between opposing bone edge convexities (fig 9a). The attachments on either side naturally settle along the tension line. Consequently, if those attachments are at the peak of each convexity, the bones will be pushed apart in a direction perpendicular to the tension force, and held there. Directional tensile stresses in the dura mater and collagen fibre orientations have been found.9,18-20 For example, symmetrical fibre orientations in the temporal regions were observed to be 6.3 degrees +/- 0.8 degrees in respect to the sagittal suture.18 At a different size scale, figure 9b demonstrates the same principle in a serrated suture. The serrated sutures increase the surface area between adjacent bones because of their interlocking projections, but the tension attachments holding the bones apart, as described above, would also decrease the potential for sutural compression in this model. [Since the publication of this paper, it has been shown that tensioned collagen fibres within the sutures are aligned such that they resist compression, as described here. [Jasinoski SC, Reddy BD, Louw KK, Chinsamy A. 2010 Mechanics of cranial sutures using the finite element method. Journal of Biomechanics 43:3104-3111.] In figures 9a and 9b the tension cords are causing the bones to be pushed apart. This is strange behaviour indeed, considering that tension is generally noted for pulling, and not pushing. It underlines how the non-linear relationship between stress and strain in tensegrity and biological structures could be brought about. Conflicting forces resolve themselves by taking the paths of least resistance, eventually settling into a stable and balanced state of minimal energy. However, a living organism has a field of force dynamics which are in a continuous state of flux, so that stability and balance are constantly changing (if that is not a contradiction in terms).32,47,48
Cells of the dura mater respond to brain expansion and influence bone growth, allowing the cranium to match the spatial requirements of the developing brain, whilst remaining one step ahead and retaining a certain autonomy.1-6 This position renders the vault more adaptable to other functional requirements, such as the demands of external musculo-tendinous and fascial attachments.7,21A tensegrity cranium balances its stability through all stages of development, by allowing small and incremental changes compatible with the mechanical demands of all connected structures.
3.3. Energetically efficient
Energetically efficient means it has maximum stability for a given mass of material. The geodesic dome can enclose a greater volume for minimal surface area, with less material than any other type of structure apart from a sphere. When the diameter of a sphere doubles, the surface area increases 4 fold and the volume increases 8 fold, which makes it materially very efficient. The entire structure of the model neurocranium resembles a sphere-like geodesic dome (fig. 3a), with a dural ‘skin’ under tension and bones enmeshed as an endoskeleton. In mechanical terms, a tensegrity structure cannot be anything other than in a balanced state of minimal energy throughout.35,45
3.4. Integration
In a tensegrity structure, a change in any one tension or compression element causes the whole shape to alter and distort, through reciprocal tension, distributing the stresses to all other points of attachment.29,30,32,35-41 In this model, the occiput is fixed at the condyles whilst the sphenoid exerts an elastic compression through the spheno-basilar synchondrosis. Apart from this, the frontal, ethmoid, sphenoid, occiput, temporals and parietals do not make direct contact with each other at any point (‘discontinuous compression’), and are suspended all around (‘continuous tension’) (fig. 8). It has been known for a long time that cranial base dysmorphology may be fundamental to the aetiology of premature suture closure.1,2 The cartilage growth plates in the chondrocranium have been shown to respond to mechanical stresses, although normally the spheno-basilar region is the only one to remain metabolically active for very long after birth, and remains so until adolescence.6,49,50 The dural sheets connecting across the neurocranium short cut mechanical stresses from one part to the other1 – the falx cerebri/cerebelli linking the ethmoid, frontal, parietals and occiput; and the tentorium cerebelli linking the sphenoid, temporals and occiput with the falx along the straight sinus. [The wire model shown is an extra figure, and the shapes correspond to the edges of the inner bone surfaces. All the ‘bones’ in this model remain separated because of the tensegrity configuration.]
The icosahedron has several attributes that are advantageous for modelling biological structures.35,36 A full account is beyond the scope of this paper, but a few significant points are worth mentioning. It is fully triangulated, which is the most stable of truss configurations (figure 3a); it comes closest to being spherical, with the largest volume to surface area ratio of all the regular polyhedrons – making it materially efficient; its surfaces can be divided equally into smaller triangles and the structure scaled up into higher frequencies – making it even more energetically efficient;43-45 it provides a link between close-packing in 2 and 3 dimensions; and as a fractal generator, it can polymerize into a sheet, stack in a column or helix, and create complex patterns and shapes. Fractal analysis is commonly applied to natural structures. Their formal definition is rather obtuse for the purposes of this paper, but a working definition could be: ‘A shape or pattern which evolves as it changes, reappearing in a hierarchy of different size scales’. Although the frequencies and amplitudes of the ‘wave’ curvatures seen at the bone edges in figures 9a and 9b vary, they are both examples of a fractal nature – with a similar pattern appearing at different size scales.51 Fractals are probably relevant to linking structural hierarchies throughout the body,2,32,35,36 thus making the icosahedron particularly versatile, because it also gives rise to structures with geodesic dome and tensegrity properties.
As the vault bones approximate each other, a sort of hybrid geodesic dome/tensegrity structure would provide the required rigidity for brain protection, but with the facility for micro-mobility at the sutures.1,2,15 Tensegrity in the cranium allows for flexibility during development, and whatever other functions that patent sutures might serve beyond cranial expansion.4,7,15,21 [It is likely that this explains some of the underlying mechanisms described by ‘cranial’ osteopaths; see Palpatory phenomena page] The cranial base naturally develops a geodesic structure and provides a platform from which the vault bones could expand, through tensegrity, to accommodate brain growth. If the transfer of tensional forces in the dura mater, and the suggested mechanisms illustrated in figure 9 really do form an essential part of sutural patency, an aberration in this system which leads to compressive bone contact at any point could be one step towards a rigid geodesic dome cranium.1,5,15 This may explain why cartilage sometimes appears in sutural joints.1,14
A local tensional stress generated within the cellular cytoskeleton could transfer to the extracellular matrix of the dura mater and produce effects on other cells at some distance, with structural rearrangements throughout the network. Long-distance transfer of mechanical forces between different tissues could contribute to dural development, and be responsible for spatially orchestrating bone growth and expansion.3,28,29,30,32,34,47,49,52 Similarly, an ‘aberrant’ tensile stress from elsewhere in the cranium could exert its effects on sutures some distance away, and contribute to a change in interactions between the dura mater, bones and brain, ultimately leading to premature synostosis.1,2
CONCLUSION
The tensegrity model is a novel approach to understanding how the cranial vault could retain its stability without relying on an expansive force from an underlying brain, a situation currently unresolved.1-6 Tensional forces in the dura mater [and suture] have the effect of pushing the bones apart, whilst at the same time integrating them into a single functional unit. Sutural patency depends on the separation of cranial bones throughout normal development, and the model describes how tension in the dura mater achieves this, and influences sutural phenotype. Cells of the dura mater respond to brain expansion and influence bone growth, allowing the cranium to match the spatial requirements of the developing brain, whilst remaining one step ahead and retaining a certain amount of autonomy. Tensegrity may also be an integrating mechanism in a hierarchical structure that extends from the cell to the whole organism, with complex 3D patterns the outcome of a network of interactions which feedback on each other.2,29,30,32,36-40,47,52 This provides a context for this model and could indicate a new approach to understanding the pathologies seen in the neonate.
One of the most significant aspects of biology is the efficiency with which it packs multiple functions into minimal space. This presents a conundrum in physical modelling, as any structure will inevitably be limited in its behaviour if it is incomplete or in isolation. It must be emphasized that much of the supporting evidence for this model is circumstantial, and more research is needed to verify it, but it is compatible with current understandings of cranial physiology, and has a contribution to make to a hierarchical systems approach to whole body biomechanics.
Acknowledgement
I wish to express my sincere appreciation to Nic Woodhead, Chris Stapleton and Andrea Rippe, for their contributions and thoughts during discussions in the preparation of this paper.
References
1. Moss ML. The pathogenesis of premature cranial synostosis in man. Acta Anat1959; 37:351–70.
2. Richtsmeier JT, Aldridge K, Deleon VB, Panchal J, Kane AK, Marsh JL et al; Phenotypic Integration of Neurocranium and Brain. J Exper Zool2006;306B:360–78.
3. Henderson JH, LongakerMT, Carter DR. Sutural bone deposition rate and strain magnitude during cranial development. Bone 2004;34;(2):271-80.
4. Mao JJ. Calvarial development: cells and mechanics.Current Opinion in Orthopaedics 2005;16;(5):331-7. [Review]
5. Yu JC, McClintock JS, Gannon F, Gao XX, Mobasser JP, Sharawy M. Regional differences of dura osteoinduction: squamous dura induces osteogenesis, sutural dural induces chondrogenesis and osteogenesis. Plastic and Reconstructive Surgery 1997;100;(1):23-31.
6. Jeffery N and Spoor F. Brain size and the human cranial base: a prenatal perspective. Am J Phys Anthropol 2002;118:324-40.
7. Sabini RC and ElkowitzDE. Significance of differences in patency among cranial sutures. JAm Ost Assoc 2006;106;(10):600-4.
8. Cotton F, Rozzi FR, Valee B, Pachai C, Hermier M, Guihard-Costa AM, Froment JC. Cranial sutures and craniometric points detected on MRI. Surg Radiol Anat 2005;27:64-70.
9. McLaughlin E, Zhang Y, Pashley D, Borke J, Yu J. The load-displacement characteristics of neonatal rat cranial sutures. The Cleft Palate Craniofacial J 2000;37;(6):590-5.
10. Opperman LA. Cranial sutures as intramembranous bone growth sites. Developmental Dynamics2000;219:472-85. [Review]
11. Mommaerts MY, Staels PFJ, Casselman JW. The faith of a coronal suture grafted onto midline synostosis inducing dura and deprived from tensile stress. The Cleft Palate Craniofacial J 2001;38;(5):533-7.
12. Persing J, James H, Swanson J, Kattwinkel J. Prevention and management of positional skull deformities in infants. Pediatrics 2003;112;(1):199-202.
13. Cowan CM, Quarto N, Warren SM, Salim A, Longaker MT. Age related changes in the biomolecular mechanisms of calvarial osteoblast biology affects Fibroblast Growth Factor-2 signaling and osteogenesis. JBiol Chem 2006;278;(34):32005-13.
14. Hall BK and Miyake T. All for one and one for all: condensations and the initiation of skeletal development. Bioessays 2000;22;(2):138-147. [Review]
15. Pritchard JH, Scott JH, Girgis FG. The structure and development of cranial and facial sutures. J Anat 1956;90;(1):73-86.
16. Bradley JP, Levine JP, Blewett C, Krummel T, McCarthy GJ, Longaker MT. Studies in cranial suture biology: in vitro cranial suture fusion. The cleft palate craniofacial J 1996;33;(2):150-6.
17. Bashkatov AN, Genina EA, Sinichkin YP, Kochubey VI, Lakodina NA, Tuchin VV. Glucose and mannitol diffusion in human dura mater.Biophys J 2003; 85:3310-8.
18. Jimenez-Hamann MC, Sacks MS, Malinin TI. Quantification of the collagen fibre architecture of human cranial dura mater. J Anat 1998;192:99-106.
19. Runza M, Pietrabissa R, Mantero S, Albani A, Quaglini V, Contro R. Lumbar dura mater biomechanics: experimental characterization and scanning electron microscopy observations. Anesth Analg 1999;88:1317.
20. Henderson JH, Nacamuli RP, ZhaoB, Longaker MT, Carter DR. Age-dependent residual tensile strains are present in the dura mater of rats. J Royal Soc Interface 2005;2:159–167.
21. Balboni AL, Estenson TL, Reidenberg JS, Bergemann AD, Laitman JT. Assessing age-related ossification of the petro-occipital fissure: laying the foundation for understanding the clinicopathologies of the cranial base. The Anatomical Record part A 2005;282A:38-48.
22. Greenwald JA, Mehrora BJ. Spector JA,Warren SM. Crisera FE, Fagenholz PJ. et al Regional differentiation of cranial suture-associated dura mater in vivo and in vitro: implications for suture fusion and patency.J Bone Miner Res 2000;15:2413-30.
23. Ogle RC, Tholpady SS, McGlynn KA, Ogle RA. Regulation of cranial suture morphogenesis. Cells Tissues Organs 2004;176:54-66.
24. Kallun R and Neilson EG. Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest 2003;112:1176-84.
25. Hay ED. The mesenchymal cell, its role in the embryo, and the remarkable signaling mechanisms that create it. Developmental Dynamics 2005;233;(3):706-20. [Review]
26. Byron D. Role of the osteoclast in cranial suture waveform patterning. The Anatomical Record part A: Discoveries in molecular, cellular and evolutionary biology. 2006;288A,(5):552-63.
27. Vij K and Mao JJ Geometry and cell density of rat craniofacial sutures during early postnatal development and upon in vivo cyclic loading. Bone 2006;38:722-30.
28. Mao JJ. Mechanobiology of craniofacial sutures. J Dent Res 2002;81;(12):810-6.[Review]
29. Ingber DE. Tensegrity I. Cell structure and hierarchical systems biology. J Cell Sci 2003;116:1157-73.
30. Ingber DE. Tensegrity II. How structural networks influence cellular information processing networks. J Cell Sci 2003;116:1397-408.
31. Sarasa-Renedo AS and Chiquet M. Mechanical signals regulating extracellular matrix gene expression in fibroblasts. Scand J Med Sci Sports 2005;15;(4):223-30.
32. Ingber DE. Cellular mechanotransduction: putting all the pieces together again.The FASEB J 2006;20:811-27.
33. Kumar S, Maxwell IZ, Heisterkamp A, Polte TR, Lele TP, Salanga M et al Viscoelastic retraction of single living stress fibres and its impact on cell shape, cytoskeletal organization, and extracellular matrix mechanics. Biophys J 2005;90:3762-73.
34. LeDuc PR and Bellin RB. Nanoscale intracellular organization and functional architecture mediating cellular behavior. Ann BioMed Eng 2006;34;(1):102-13. [Review]
35. Fuller BB. Synergetics, explorations in the geometry of thinking. Macmillan; 1975.
36. Levin S. The importance of soft tissues for structural support of the body.Spine: state of the art reviews. Dorman,T. ed. 1995;9;(2). [see ref. 40]
37. Levin S. Putting the shoulder to the wheel: a new biomechanical model for the shoulder girdle. J Biomed Sci Instrum 1997;33. [see ref.40]
38. Levin S. The tensegrity truss as a model for spine mechanics: biotensegrity.J Mech Med Biol 2002;2:375-88. [see ref.40]
39. Levin S. A suspensory system for the sacrum in pelvic mechanics: biotensegrity. In: Vleeming A, Mooney V, Stoeckart R. editors. Chap 15: Movement, stability and lumbopelvic pain. Churchill Livingstone 2007. [see ref.40]
40. Levin S. www.biotensegrity.com Includes references: 36-39.
41. Snelson K. www.kennethsnelson.net
42. Stamenovi D and Coughlin MF. A prestressed cable network model of the adherent cell cytoskeleton. Biophysical Journal 2003;84:1328-36.
43. Weinbaum S. Mechanotransduction and flow across the endothelial glycocalyx. PNAS 2003;100;(13):7988-95.
44. Li J, Dao M, Lim CT, Suresh S. Spectrin-level modeling of the cytoskeleton and optical tweezers stretching of the erythrocyte. Biophys J 2005;88;(5):3707-19.
45. Connelly R & Back A. Mathematics and tensegrity. American Scientist 1998; 86:142-51.
46. Gordon JE. Structures, or why things don’t fall down. Penguin 1978.
47. Ingber DE. Mechanical control of tissue morphogenesis during embryological development. Int J Dev Biol2006;50:255-66.
48. Van Workum K and Douglas JF. Symmetry, equivalence, and molecular self-assembly. Physical Reviews 2006;73:031502.
49. Wang X and Mao JJ. Chondrocyte proliferation of the cranial base cartilage upon in vivo mechanical stresses. Dent Res 2002;81;(10):701-5.
51. Yu JC, Wright RL, Williamson MA, Braselton JP, Abell ML. A fractal analysis of human cranial sutures. The cleft palate craniofacial J 2003;40;(4):409-15.
52. Nelson CM, Jean RP, Tan JL, Liu WF, Sniadecki NJ, Spector AA et al (2005) Emergent patterns of growth controlled by multicellular form and mechanics. PNAS 2005;102;(33):11594-9.
